 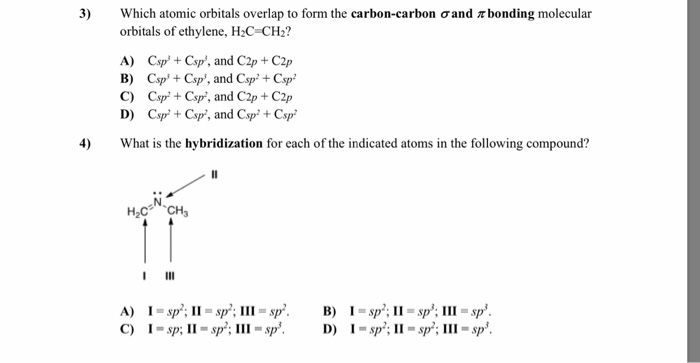 
The numbers in front of the MO's differentiate the orbitals with the same symmetry. The MO's are qualitatively ordered based on the number of nodes that are found when you combine the SLAC's. The electron configuration of this excited state suggests a molecule with three mutually perpendicular bonds based on the orbitals of the carbon atom having. Orbits are replaced with atomic orbitals, volumes of space where the. Now, these sp hybridized orbitals of the carbon atom overlap with two p orbitals of the. Excited state is an unstable, higher energy state of an atom. Finally, the 2px orbital remains unchanged because there are no other SLAC's with b1 symmetry. CH2 C CH2 Indicate sigma and pi bonds in the above molecule. The 2py (b2 symmetry) orbital combines with the b2 combination of the hydrogen atoms to form two MO's. Moreover, the Valence Shell Electron Pair Repulsion (VSEPR) theory, says the molecular geometry of a molecule is trigonal planar. In the above example, the 2s and 2pz orbitals on oxygen have a1 symmetry, and they combine with the a1 combination of hydrogen 1s orbitals to form three MO's with a1 symmetry. The CH2O is a tetra atomic molecule where the bond angles for the hydrogen-carbon-hydrogen (H-C-H) and hydrogen-carbon-oxygen (H-C-O) are 116 and 122 and the structure is bent shaped. H1 is positive and H2 is negative or vice versa).ĪO's that have the same symmetry can combine to form MO's with that symmetry label. The maximum number of atomic orbitals associated with a principal quantum. The a1 combination is when the phase of the wavefunction is the same and the b2 combination is when the phase is opposite (i.e. Structure of Atom Chemistry Practice questions, MCQs, Past Year Questions. For example, the 2py orbital is in the plane of the nuclei and so it is symmetric with respect to E and σ' but anti-symmetric with respect to C2 and σ and has b2 symmetry from the C2V character table.įor the hydrogens, you have to consider the combination of both 1s orbitals at the same time. You can determine by symmetry label for the AO's by operating on the orbital with each of the symmetry operators in the C2V group (E, C2, σ, σ'). Molecular orbitals for three-carbon systems Organic Chemistry II 13.3. Each of the atomic orbitals (AO's) on carbon (left) and the combination of $1s$ orbitals on the hydrogen atoms (right) has a symmetry label that corresponds to one of the irreducible representations of the point group. and line-bond structure CH2CHCHCH2 tells: buta part. A molecule of acetylene (C2H2) has a geometry and a molecular dipole moment that is. Rotation about the carbon-carbon bond axis disrupts the overlap of the two carbon p orbitals forming the pi bond. The first question to always ask when you are trying to build an MO diagram is what point group does my molecule fall into? In this case, the $\ce$ molecule has C2V symmetry (like the water example shown above). Distribution and Frontier Molecular Orbitals of Butadiene: General Features for Diels-Alder. In the case of CH2CH2, the carbon-carbon bond is a double bond with both a sigma and pi bond present. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
